Spectral Analysis and In Vitro Biological Activity of Cu(II) Complex with Tridentate ONO Schiff Base Ligand
Abstract
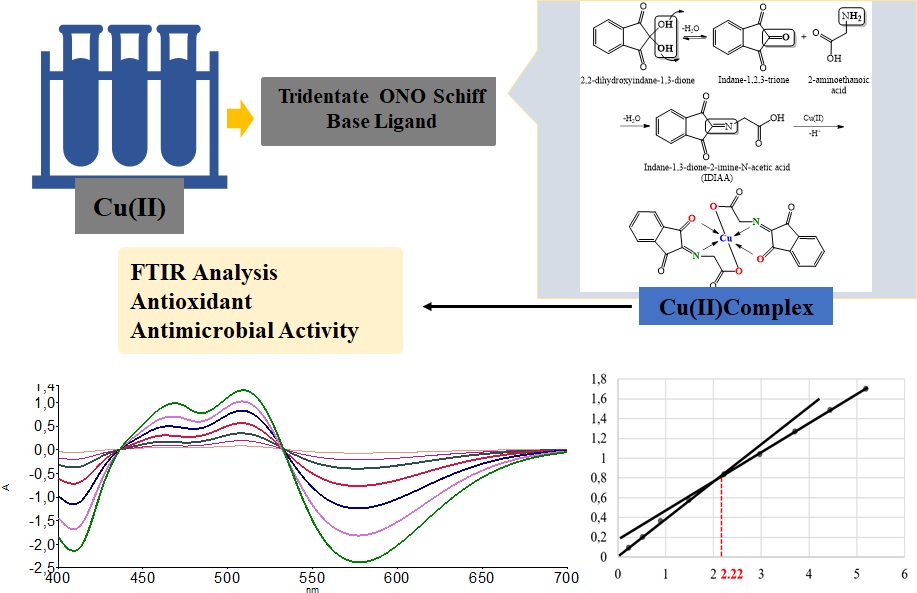
In this study, metal complex of Copper(II) with a Schiff base derived from 2,2-dihydroxyindane-1,3-dione and 2-aminoethanoic acid were synthesized. The product are characterized by spectral methods. The antimicrobial activity was tested on reference bacterial strains and the antioxidant capacity was analyzed by using the DPPH and FRAP methods. The spectral data indicates that the Schiff base coordinates the Copper(II) as a tridentate ONO donor ligand. The compounds showed weaker antimicrobial activity on certain tested microorganisms. In vitro testing of antioxidant activity showed a significant reducing ability of the complex, as well as inhibitory activity against DPPH radicals.
References
[1] Wail, A.Z., Abbas, A.S.A., Young, G.K., Separation Science and Technology, 2017, 52(6), 1052-1069.
[2] Sani, U., Na’ibi, H.U., Dailami, S.A., Nigerian Journal of Basic and Applied Science, 2017, 25(1), 81-88.
[3] Dhar, D.N., Taploo, C.L., Journal of Scientific and Industrial Research, 1982, 41(8), 501-506.
[4] Silva, C.M., Silva, D.L., Modolo, L.V., Alves, R.B., Resende, M.A., Martins, C.V.B., Fátima, A., Journal of Advanced Research, 2010, 2(1), 1-8.
[5] Bader, N.R., RASAYAN Journal of Chemistry, 2010, 3(4), 660-670.
[6] Fátima, A., Pereira, C.P., Olímpio, C.R.S.D.G., Oliveira, B.G.F., Franco, L.L., Silva, P.H.C., Journal of Advanced Research, 2018, 13, 113-126.
[7] Patil, U., Khan, A., Nagarsekar, A., Mandewale, M., Yamgar, R., Oriental Journal of Chemistry, 2018, 34(6), 2796-2805.
[8] Kafi-Ahmadi, L., Marjani, A.P., South African Journal of Chemistry, 2019, 72, 101-107.
[9] Sobola, A.O., Watkins, G.M., Brecht, B., Journal of the Serbian Chemical Society, 2018, 83(7-8), 809-819.
[10] El‐Halima, H.F.A., Omar, M.M., Mohamed G.G., Sayed, M.A.E., European Journal of Chemistry, 2011, 2(2), 178‐188.
[11] Naeimi, H., Nazifi, Z.S., Amininezhad, S.M., Amouheidari, M., The Journal of Antibiotics, 2013, 66, 687-689.
[12] Almarhoon, Z.M., Al-Onazi, W.A., Alothman, A.A., Al-Mohaimeed, A.M., Al-Farraj, E.S., Journal of Chemistry, 2019, Article ID 8152721, 14 pages.
[13] Kajal, A., Bala, S., Kamboj, S., Sharma, N., Saini, V., Journal of Catalysts, 2013, Article ID 893512, 14 pages.
[14] Horozić, E., Suljagić, J., Husejnagić, D., Hasić, N., Bratovčić, A., Journal of Engineering & Processing Management, 2019, 11(2), accepted.
[15] Sztanke, K., Maziarka, A., Osinka, A., Sztanke, M., Bioorganic & Medicinal Chemistry, 2013, 21(13), 3648-3666.
[16] Utreja, D., Singh, S., Kaur, M., Current Bioactive Compounds, 2015, 11(4), 215-230.
[17] Horozić, E., Suljagić, J., Suljkanović, M., American Journal of Organic Chemistry, 2019, 9(1), 9-13.
[18] Derebe, M.G., Raju V.J.T., Negussie R., Bulletin of the Chemical Society of Ethiopia, 2002, 16(1), 53-64.
[19] Yoe, J.H., Jones, L., Industrial and Engineering Chemistry, Analytical Edition, 1944, 16(2), 111-115.
[20] Horozić, E., Zukić, A., Kolarević, L., Bjelošević, D., Ademović, Z., Šarić-Kundalić, B., Husejnagić, D., Kudumović, A., Hamzić, S., Technics Technologies Education Management, 2019, 14(1), 14-19.
[21] Benzie, I.F.F., Strain, J.J., Methods in Enzymology, 1999, 299, 15-27.
[22] CLSI MO2-A11. Performance Standards for Antimi-crobial Disk Susceptibility Tests, Aproved Standard, eleventh edition. Clinical and Laboratory Standards Institute (CLSI) 2012; ISBN 1-56238-986-6.
[23] Ouedrhiri, W., Balouiri, M., Bouhdid, S., Sandrine, M., Fouad, O.C., Taleb, M., Greche, H., Industrial Crops and Products, 2016, 89, 1-9.
Refbacks
- There are currently no refbacks.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.