Synthesis and Characterization of PVDF-LiBOB Electrolyte Membrane with ZrO2 as Additives
Abstract
The electrolyte membrane serves as ions medium transport and as a separator between the anode and cathode in lithium ion battery. The polymer used for the electrolyte membrane must have sufficiently high mechanical strength to withstand the pressure between the anode and cathode, a thin size and has a chemical and thermal stability.
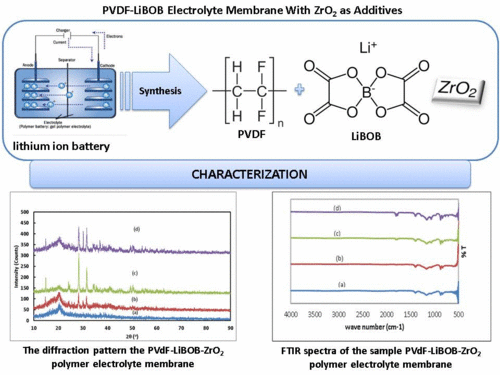
Polymer electrolyte membrane of Lithium bisoxalate Borate(LiBOB) salt with PVdF as matrix polymer and the additive is ZrO2 has been fabricated. The method used is a doctor blade. The concentration of the additive is varied. The membranes were characterized using FT-IR, XRD, SEM and EIS. XRD analysis showed that the crystallinity index increases with the addition of ZrO2. The presence of functional groups bewteen Lithium salts and polymer interaction shown by FTIR analysis. The morphology of the membrane surface was shown by SEM analysis. SEM image and mapping show the morphology of the membrane have typical porous layer. The electrical conductivity increases with additions of ZrO2.References
Larush-Asraf, L., Biton, M., Teller, H., Zinigrad, E. and Aurbach, D., J. Power Sources, 2007, 174, 400–407.
[2] J. Zhang, B. Sun, X. Huang, G. Wang, Conference Proceedings APEnergy, 2014.
[3] Wu, X.-L. Xin, S. Seo, H., Kim. J., Guo, G., and Lee, J., Solid State Ion., 2011, 186, 1–6.
[4] Li, Y.H. Wu, X.L., Kim, J.H., Xin, S., Su, J., Yan, Y., Lee, J.S. and Guo, Y.G, J.Power Sources, 2013, 244, 234–239.
[5] Wang, X. Gong, C., He, D., Xue, Z., Chen, C., Liao, Y. and Xie, X., J. Memb. Sci., 2014, 454, 298–304.
[6] Li, W., Xing, X., Li, Y., Yang, G. and Xu, L., Electrochim. Acta, 2013, 112, 183–190.
[7] Nunes-Pereira, J., Costa, C. M. and Lanceros-Méndez, S., J. Power Sources, 2015, 281, 378–398.
[8] Zhang, R., Y. Chen, R. Montazami, Materials, 2015, 8, 2735–2748.
[9] An, S.J., J Li, C Daniel, D Mohanty, S Nagpure, D. L Wood, Carbon, 2016, 105, 52–76.
[10] Weng, B., F. Xu, M. Alcoutlabi, Y. Mao, K. Lozano, Cellulose, 2015, 22, 1311–1320.
[11] A.D. Epifanio, M.A. Navarra, F.C. Weise, B. Mecheri, J. Farrington, Chem Mater, 2010, 22, 813–821.
[12] M.M. Navarra, C.Abbati, B.Scrosati, J. Power Sources, 2008, 183(1), 109–113.
[13] Kobayasyi, Y., S. Seki, A. Yamanaka, H. Miyashiro, Y. Mita, T. Iwahori, J. Power Souces, 2005, 146, 719–722.
[14] Segal L, Creely, J.J, Martin, A.E, Conrad C.M, Text. Res. J., 1962, 29, 786–794.
[15] Mano, J. F., Costa, a M. dan Schmidt, V. H., J. Macromol. Sci. Phys., 2001, 40, 517–527.
[16] Abdullah, M., Lenggono, W., dan Okuyama, K. Encyc. Nanosci. And Nanotechnol., 2004, 8, 731–762
[17] Aravindan V, Vickraman P, J. Phys D: Apply. Phys, 2007, 40, 6754–6759.
[18] J. Y. Kim, D. Y. Lim, Energies, 2010, 3, 866–885.
[19] Wigayati, E.M., T. Lestariningsih, A. Subhan, C. R. Ratri, I. Purawiardi, Ionics, 2015, 22(1), 43–50.
[20] E. M. Wigayati, C. R. Ratri, I. Purawiardi, F.Rohman, T. Lestariningsih, Indones. J. Chem, 2015, 15(3), 242–247.
Refbacks
- There are currently no refbacks.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.